
AR Biotech Consulting accelerates your research to bring biologics from concept to CMC-ready reality.
About
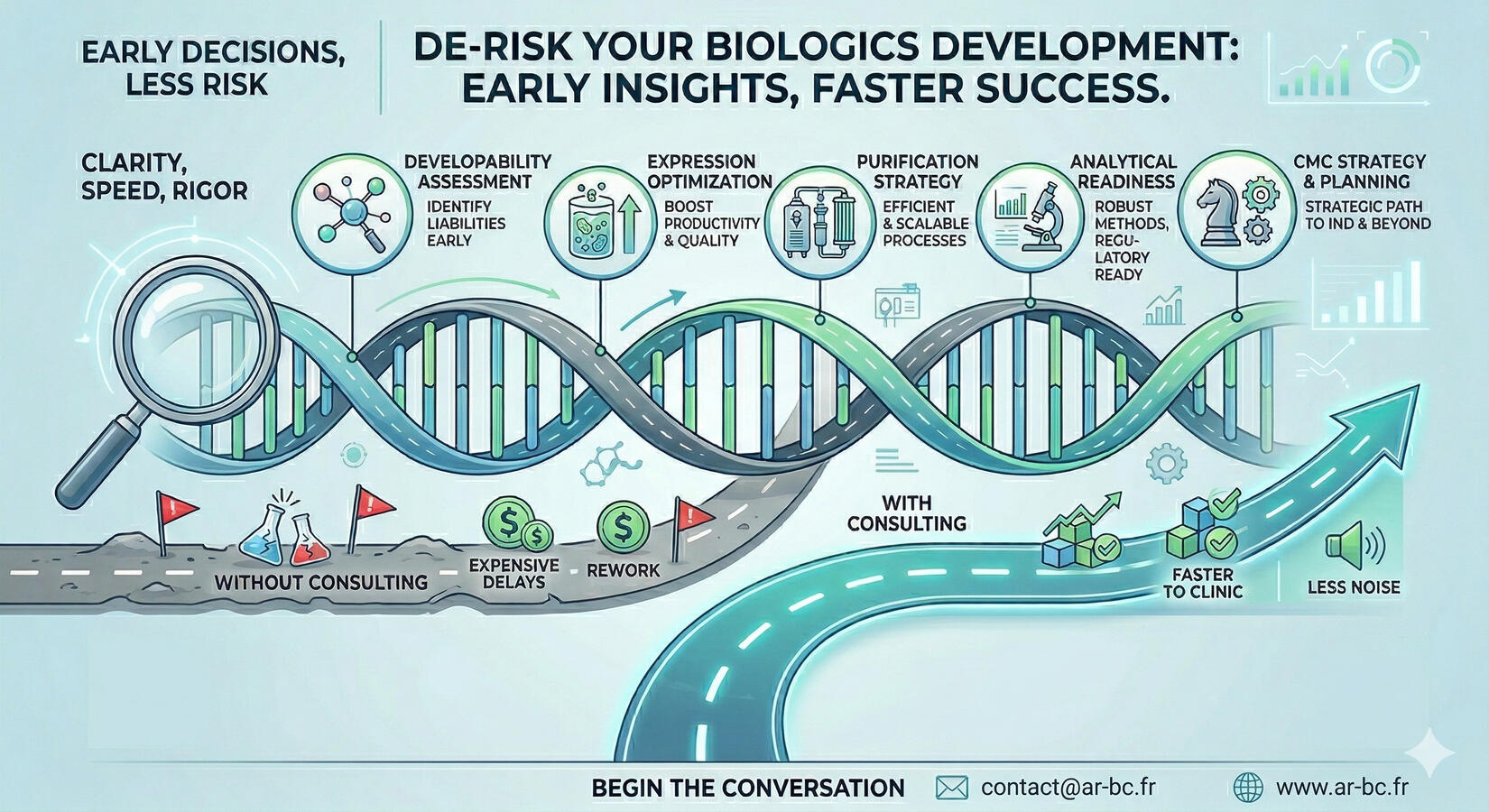
When a biologics program needs clarity, speed and scientific rigor, the difference often comes from the quality of the early decisions.The goal is to help your team de-risk development before problems become expensive delays. That means focusing on the critical points that shape success: developability, expression, purification, analytical and CMC strategy.Core expertise includes:
- Developability assessment
- Expression optimization
- Purification strategy and troubleshooting
- Analytical readiness and method strategy
- CMC planning and risk management
- Tech transfer support
- Early-stage biologics de-riskingThis kind of work is most valuable when the margin for error is small and the stakes are high. Whether your program is preparing for scale-up, tech transfer or IND-enabling work, the objective is the same: identify risks early, prioritize what matters and move forward with a clearer path.You get more than technical support. You get an experienced, independent perspective that helps your team make better decisions, faster — with less noise, less rework and more confidence in the next step.If your program needs a senior scientific partner who understands both the technical detail and the broader development picture, this is where that conversation begins.
Contact
For project inquiries, collaborations or consulting support in biologics development and CMC, please get in touch[email protected]